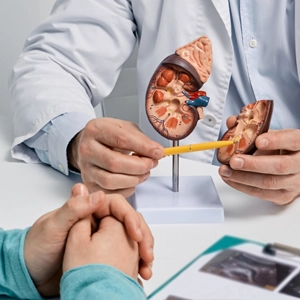
Health Canada Approves Ipsen’s CABOMETYX™ (cabozantinib) for the First-Line Treatment of Adults with Advanced Renal Cell Carcinoma
Source: Ipsen Biopharmaceuticals Canada Inc.
CABOMETYXTM demonstrated a statistically significant, 52 per cent reduction in the risk of progression or death compared with sunitinibi, giving Canadian patients with intermediate or poor risk a new oral first-line treatment option in their fight against advanced kidney cancer
MISSISSAUGA, ON, Oct. 15, 2019 /CNW/ – Ipsen Biopharmaceuticals Canada Inc. today announced the Health Canada approval of CABOMETYXTM (cabozantinib) 20, 40, 60 mg for the first-line treatment of adults with advanced renal cell carcinoma (aRCC), the most common type of kidney cancer.ii
“The Health Canada approval helps strengthen our treatment arsenal for patients with advanced renal cell carcinoma,” says Dr. Anil Kapoor, Professor of Oncology and Surgery at the Juravinski Cancer Centre at McMaster University and Chair of the National Kidney Cancer Research Network of Canada. “It adds a new option for first line treatment that demonstrated superior clinical efficacy when compared to sunitinib, a current standard of care.”
On average, 7,200 Canadians are diagnosed with kidney cancer each year,iii and the incidence is growing.iv Unfortunately, many kidney cancers are diagnosed at a later stage, or progress to later stages. It is estimated that 1,900 Canadians will die from kidney cancer in 2019, highlighting the need for new treatment options for patients and caregivers.v
“A critical goal for many patients is to prolong survival while maintaining quality of life,” says Stephen Andrew, Executive Director of Kidney Cancer Canada. “Today’s announcement is important because it gives patients more options in the fight against the disease.”
This approval was based on the CABOSUN trial which demonstrated that CABOMETYXTM significantly prolonged progression-free survival (PFS) compared to sunitinib in treatment naïve aRCC patients with intermediate or poor-risk disease.vi CABOMETYXTM is the first and only monotherapy to demonstrate superior clinical efficacy over sunitinib in treatment-naïve aRCC patients with intermediate or poor-risk.
“We come to work every day with the goal of providing hope for patients with difficult-to-treat diseases, and we are thrilled that this expanded indication for CABOMETYXTM widens the scope of patients we can help,” says Ed Dybka, General Manager, Ipsen Canada. “Today’s announcement further demonstrates the efficacy and safety of CABOMETYXTM and reinforces our commitment to expanding its potential across different indications.”
NOTE FROM KIDNEY CANCER CANADA
Although Health Canada has approved cabozantinib in first-line, this is the first step in a process before this indication is approved for use in Canada. This indication now has to be evaluated by Health Technology Process to see if clinicians, payers (private and government) believe cabozantinib benefits patients in the first-line setting over therapies currently in use. Kidney Cancer Canada – Cancer du rein Canada submits it’s recommendation and position during this process with an evidence based decision.
Cabozantinib is recommended in Canada for use in subsequent lines of therapies. However, as of today, we wait for the company and government to complete and announce the result of their price negotiation, which would make it available to patients in Canada.
About the CABOSUN Trial
CABOSUN was a randomized, open-label, active-controlled phase II study that enrolled 157 patients with aRCC who were intermediate or poor-risk according to International Metastatic RCC Database Consortium (IMDC) criteria as determined by investigator assessment. Patients with previously untreated, locally advanced or metastatic RCC with a clear cell component were randomized (1:1) to receive CABOMETYXTM 60 mg once daily (N=79) or sunitinib 50 mg once daily, four weeks on, followed by two weeks off (N=78); The primary efficacy endpoint was progression free survival (PFS). Secondary endpoints included overall survival (OS) and objective response rate (ORR).vii
In the trial, CABOMETYXTM demonstrated a clinically meaningful and statistically significant reduction in the rate of disease progression or death as measured by PFS. Median PFS for CABOMETYX in the Independent Review Committee (IRC) assessment was 8.6 months versus 5.3 months for sunitinib (HR=0.48, 95% CI, p=0.0008).viii The most common adverse reactions in ≥ 10% of CABOMETYXTM treated patients were diarrhea (73%), hypertension (67%), fatigue (64%) and increased levels of aspartate aminotransferase (60%).ix
About CABOMETYXTM (cabozantinib)
CABOMETYXTM is the first multi-targeted tyrosine kinase inhibitor (TKI) to target VEGFR, MET and AXL pathways that are involved in tumour progression in RCC. It works by blocking the action of proteins called receptor tyrosine kinases (RTKs). RTKs are involved in cell growth and the development of new blood vessels. These proteins can be present in high amounts in cancer cells. By blocking their action, CABOMETYXTM can slow down how fast the tumour grows, help to block the blood supply that the cancer needs and may increase the length of time before the cancer gets worse.x
Following a priority review by Health Canada, CABOMETYXTM was first approved in Canada in September 2018 for the treatment of adult patients with aRCC who had received prior vascular endothelial growth factor (VEGF)-targeted therapy.xi
About Ipsen
Ipsen Biopharmaceuticals Canada Inc., the Canadian affiliate of Ipsen, is headquartered in Mississauga, Ontario with established operations since October 2015. For more information on Ipsen Biopharmaceuticals Canada Inc. visit www.ipsen.ca. Ipsen is a global specialty-driven biopharmaceutical group focused on innovation and specialty care. The group develops and commercializes innovative medicines in three key therapeutic areas – Oncology, Neuroscience and Rare Diseases. Its commitment to Oncology is exemplified through its growing portfolio of key therapies for prostate cancer, neuroendocrine tumors, renal cell carcinoma and pancreatic cancer. Ipsen’s R&D is focused on its innovative and differentiated technological platforms located in the heart of the leading biotechnological and life sciences hubs (Paris-Saclay, France; Oxford, UK; Cambridge, US). The Group has about 5,700 employees worldwide. Ipsen is listed in Paris (Euronext: IPN) and in the United States through a Sponsored Level I American Depositary Receipt program (ADR: IPSEY). For more information on Ipsen, visit www.ipsen.com.
i Choueiri TK, Hessel C, Halabi S, et al. Cabozantinib versus sunitinib as initial therapy for metastatic renal cell carcinoma of intermediate or poor risk (Alliance A031203 CABOSUN randomized trial): Progression‐free survival by independent review and overall survival update. Eur J Cancer. 2018;94:115‐25.
ii Mayo Clinic, Kidney Cancer Overview, https://www.mayoclinic.org/diseases-conditions/kidney-cancer/symptoms-causes/syc-20352664, Last accessed October 1, 2019.
iii Canadian Cancer Society. Kidney Cancer Statisticshttps://www.cancer.ca/en/cancer-information/cancer-101/canadian-cancer-statistics-publication/?region=on , Last accessed October 1, 2019
iv Cancer Care Ontario. https://www.cancercareontario.ca/en/cancer-facts/kidney-cancer-incidence-rates-rising, Accessed August 22, 2019.
v Canadian Cancer Society. Kidney Cancer Statisticshttps://www.cancer.ca/en/cancer-information/cancer-101/canadian-cancer-statistics-publication/?region=on , Last accessed October 1, 2019
vi CABOMETYXTM Product Monograph. Ipsen Biopharmaceuticals Canada Inc. November 12, 2018
vii CABOMETYXTM Product Monograph. Ipsen Biopharmaceuticals Canada Inc. November 12, 2018
viii CABOMETYXTM Product Monograph. Ipsen Biopharmaceuticals Canada Inc. November 12, 2018
ix CABOMETYXTM Product Monograph. Ipsen Biopharmaceuticals Canada Inc. November 12, 2018
x CABOMETYXTM Product Monograph. Ipsen Biopharmaceuticals Canada Inc. November 12, 2018
xi Health Canada, https://hpr-rps.hres.ca/reg-content/regulatory-decision-summary-detail.php?lang=en&linkID=RDS00492, Accessed August 23, 2019




























































Chuck Mac Donald
June 15, 2020 at 12:04 pmAm wondering if I could get more info.
Can I take part in the trial
Any further information you can get to me would be great
Thanks
Chuck
Susan Nemeth
June 15, 2021 at 3:38 pmI have Neurofibromatosis and I was told that they are treating individuals with Neurofibromatosis with this drug to shrink their tumors. I am very interested in getting this medication. As I suffer severe pain throughout my body from these tumors. They are also very disfiguring and cause me to have low self-esteem and anxiety when going out into the community. I also now suffer PTSD because of all the negative experiences I have suffered. Please send me any information you can about this medication. Thank you
Susan